Explain Different Type of Hydrogen Bond With Example
O-nitrophenol and salicylic acid are examples of intramolecular hydrogen bonding. This type of covalent bond is formed whenever there is an equal share of electrons between atoms.
Hydrogen Bond Definition Types And Examples
Nylon is used in many fabrics as well as other everyday items like toothbrushes and hairbrushes.

. The intramolecular hydrogen bond is the hydrogen bond formed between the hydrogen atoms and electronegative atoms like nitrogen oxygen and sulfur within the same molecule. Explain different types of hydrogen bond giving examples. Therefore hydrogen bonding arises in water molecules due to the dipole-dipole interactions between the hydrogen atom of one water molecule and the oxygen.
Lower alcohols are more soluble in water because of the hydrogen bonding between a molecule of alcohol and water. Depending on the occurrence of the hydrogen bonding hydrogen bonds are of two types. Hydrogen bonding in water leads to the molecular association of.
Vander waals forces are weak forces and exist in non -polar compounds and noble gases As compared to hydrogen bonds they are weak in nature because of strong dipole -dipole interaction in hydrogen bonding. This pictures shows examples of chemical bonding using Lewis dot notation. It is formed when a hydrogen atom is in between the two highly electronegative F O N atoms present within the same molecule.
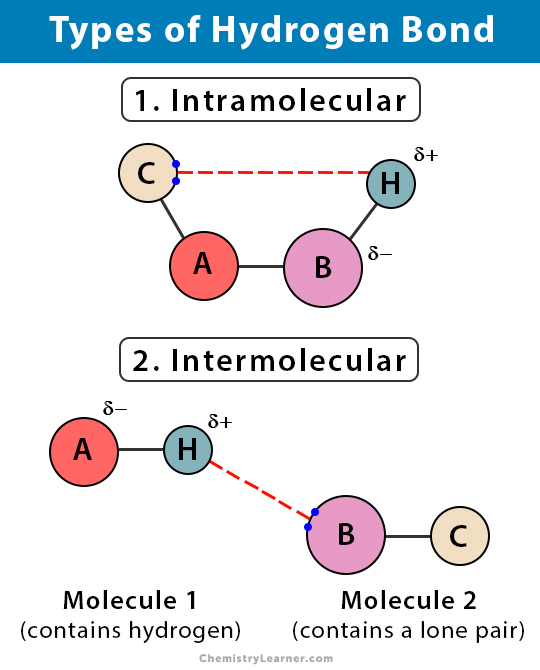
Ii Intramolecular H-bond eg o-nitrophenol. In this case the hydrogen atom interacts with electronegative fluorine hydrogen or oxygen. Electrostatic force of attraction that exists between hydrogen atom of one molecule and electronegative atom of another molecule is called intermolecular hydrogen atom.
The hydrogen bonds in the molecules of this man-made material are responsible for the crystallization of the material in the amide repeat unit. This is a. Example molecules forming hydrogen bonding as a result of an unbalanced electrostatic potential.
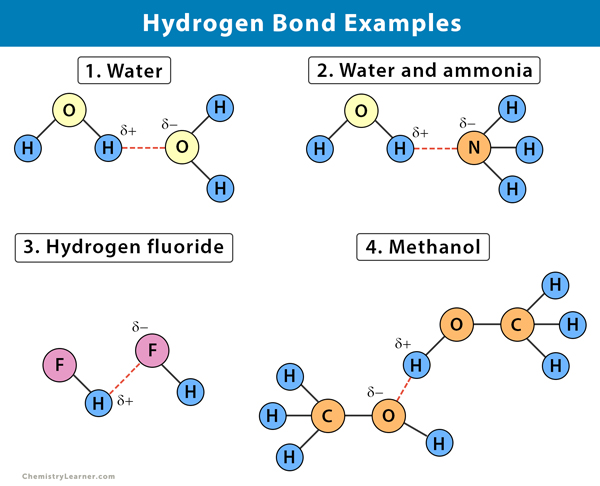
Hydrogen bonding in HF. This type of bonding can be found in Hydrofluoric acid HF water H2O etc. Water methyl alcohol ethyl alcohol and sugar are examples of intermolecular hydrogen bonding.
Bonds especially covalent bonds are often represented as lines between bonded atoms. Intermolecular hydrogen bonding This type of bond formation takes place between the distinct molecules of equal or specific compounds. The type of hydrogen bonding which takes places within the molecule is called intramolecular hydrogen bonding.
Examples include urea and polyurethane and the natural polymer cellulose. Types of Hydrogen Bonding Intermolecular Hydrogen Bonding. In hydrogen fluoride HF the positive end of one dipole attracts the negative end of another similar dipole.
Properties of Hydrogen Bond. Hydrogen bond formed between hydrogen atom of a molecule and highly electronegative atoms of the same molecule is known. Intramolecular hydrogen bonding This type of bond formation happens when the hydrogen atom lies in between the two electronegative elements gift inside the same molecule.
For example in water molecules H 2 O hydrogen is covalently bonded to the more electronegative oxygen atom. Polymers that contain carbonyl or amide groups can form hydrogen bonds. Ethanol and other alcohols contain hydrogen bonds between hydrogen and oxygen.
Type of hydrogen bond. What is hydrogen bond explain the different types of hydrogen bond with examples. The hydrogen bonding which takes place within a molecule itself is called.
Hydrogen bonds may form between atoms within a molecule or between two separate molecules. Hydrogen bonding in water. Hydrogen bond formed between two different polar molecules of same or different substances is known as intermolecular hydrogen bond.
As an instance- hydrogen bonding in water and alcohol. Therefore the melting and boiling points of such compounds are often high. Water ammonia and hydrogen fluoride are examples of such types of molecules.
Diploe-Dipole Interaction In a covalent bond if there is an electronegativity difference between the two atoms then the electrons get pulled more towards the electronegative atom and it has a higher electron density. Hydrogen bonds are of two types i Intermolecular H-bond eg HF H 2 O etc. Therefore more energy is required to separate the molecules of these compounds.
1Intermolecular Hydrogen bonding When hydrogen bonding takes place between different molecules of the same or different. Nylon a synthetic polymer famous for its stretchy qualities is another hydrogen bond example. Acetylene has a triple bond a special type of covalent bond that will be.
A hydrogen bond is weaker than an ionic bond or a covalent bond but stronger than van der Waals forces. 1 Intermolecular hydrogen bond. Intermolecular hydrogen bonding occurs in HF and H2O.
Intermolecular Hydrogen Bonding. When hydrogen bonding takes place between different molecules of the same or different. Hydrogen and carbon are not bonded while in water there is a single bond between each hydrogen and oxygen.
These molecules are associated together to form the cluster HF n. Compounds which have intermolecular hydrogen bonding their molecules are attracted to each other due to hydrogen bonding. Hydrogen bonding in these molecules increases their tensile strength and melting point.
Because compounds with hydrogen bonds between different molecules have a higher boiling point they are less volatile. Types of hydrogen bonds. 2 Intramolecular hydrogen bond.
One of the atoms is hydrogen while the other may be any electronegative atom such as oxygen chlorine or fluorine. HF H20 NH3 etc.
Hydrogen Bond Definition Types And Examples

Define Hydrogen Bond Explain Different Types Of Hydrogen Bond Giving Examples
Hydrogen Bonding Properties Effects Types Examples Of Hydrogen Bond
Comments
Post a Comment